Bertrand Clinical Label
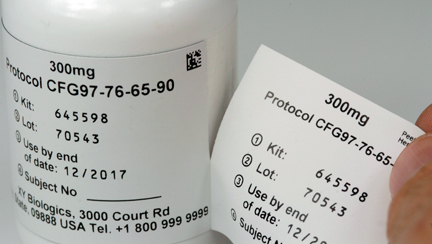
USE – This two part label construction is used to provide an identifiable label that will be applied to the patient's case report by the provider or doctor.
CONSTRUCTION – It consists of paper or film material with a pressure sensitive adhesive. This construction uses a perforation on top of the label and face slits in the liner to allow a section to flag off with its adhesive remaining covered. The second label (or flagged section) is separated and affixed to the patient's paperwork by the provider or doctor. There are perforations separating each label.
AVERAGE LEAD TIME WITH STATIC TEXT – 7-10 days from approved proofs.
VARIABLE PRINT - Add 3-4 days from approved variable proofs.
OPTIONS
2-part labels have a removable segment that can be peeled off and attached to documentation, dispensing records, or patient files, providing an easy audit trail for clinical trials.
2-part labels ship in approximately 10–12 working days from approved proof with materials in stock.
Yes. Variable codes such as kit numbers and barcodes can be printed on both segments of 2-part labels.
Yes. We have extensive experience producing 2-part labels for blinded and double-blinded clinical studies with strict unblinding controls.
Yes. We can configure the removable panel to carry patient ID, randomization codes, or other study-specific information.